SUITABILITY OF HYDROCARBON AND CARBON MONOXIDE MEASUREMENT
AS COMBUSTION INDICATORS IN CEMENT KILNS
Operations Consultant - Gossman Consulting, Inc. Rex Jameson
Corporate Production Engineer - Holnam Inc. Carlos Buckelew
Plant Manager - Lone Star Industries Inc. Henry Winders
Director of Environmental Affairs - Continental Cement Co. Richard Parker
Manager-Fuels Systems - Safety-Kleen Corporation Marcia Williams
President-Williams & Vanino, Inc. David Gossman
President - Gossman Consulting, Inc.
Presented at the AWMA International Specialty Conference on Waste
Combustion in Boilers and Industrial Furnaces March, 1993
ABSTRACT
Carbon monoxide (CO) and hydrocarbons (HC) are established combustion indicators in hazardous waste incinerators. Incinerator logic was subsequently used in developing the Boiler and Industrial Furnace (BIF) rules. Cement kilns and incinerators are significantly different devices. Elevated CO levels in cement kilns have been demonstrated to occur independent of combustion conditions. HC emissions have also resulted in cement kilns due to naturally occurring hydrocarbons in raw materials. This phenomenon is also independent of combustion conditions, since raw material hydrocarbons never reach the combustion zone. In addition, CO and HC levels vary significantly under stable kiln operating conditions, sometimes exhibiting an inverse relationship. These and other factors are examined and discussed.
INTRODUCTION
Test after test continues to demonstrate that neither HC nor CO are reliable indicators of combustion conditions in cement kilns. In some cases, HC and CO actually exhibit an inverse relationship. The following issues are critical in reconciling the BIF assumptions with actual cement kiln combustion.
- Current HC and CO limits are both arbitrary and based upon incinerator data.
- Cement kilns and incinerators are significantly different devices. - Raw material in cement kilns is necessary to produce cement.
- CO levels vary significantly, a phenomenon unrelated to combustion conditions. - Setting a CO baseline based upon current methodology is consequently not meaningful.
- Elevated CO levels occur in virtually all cement kilns independent of combustion conditions for the following reasons: - Calcination, which releases CO2 and small quantities of CO, is necessary for cement production.
- HC levels vary significantly irrespective of combustion conditions or CO levels. - Measuring HC has no relevance toward controlling combustion conditions.
- HC emissions occur because of natural hydrocarbon sources (i.e. kerogens) in raw materials which are unrelated to combustion conditions.
- HC cannot be used as an alternative to CO because tests under starved oxygen conditions have demonstrated negligible effects on HC. Monitoring HC fails to detect poor combustion conditions.
- It is possible to establish process control indicators which are conservatively protective relative to preventing unsafe levels of PICs. - These indicators, specific to cement kilns, cannot be applied to incinerators any more successfully than the current BIF indicators, based upon incinerators, can be applied to cement kilns.
- Gas flows are counter current in a cement kiln.
- There are longer retention times and higher temperatures in cement kilns.
- Decomposition of carbon dioxide into carbon monoxide and oxygen occurs due to the elevated temperatures in a cement kiln.
- Decomposition of carbon dioxide into carbon monoxide and oxygen may occur due to moisture and metallic catalysts, such as iron.
A CEMENT KILN IS NOT AN INCINERATOR
The EPA promulgated the Boiler and Industrial Furnace Regulations (BIF) on February 21, 1991 with the intent of "expanding controls on hazardous waste combustion to regulate air emissions from the burning of hazardous waste in boilers and industrial furnaces." "In addition, the rule controls emissions of products of incomplete combustion (PICs) by limiting flue gas concentrations of carbon monoxide (CO), and where applicable, hydrocarbons (HC) to ensure that the device is operated under good combustion conditions."
Clearly, the monitoring of CO and HC required under BIF has resulted due to EPA intention of preventing combustion related PICs. This is made particularly clear as regards requirements for "Monitoring in the By-Pass Duct of a Cement Kiln" whereby potential main stack PICs are not addressed at all. These PICs are not addressed since presumably they are recognized as being raw material related, and as cited in this section "...gases in the bypass duct are representative of the combustion of waste in the kiln." (56FR7159, left-hand column)
Consider also, that the 100 ppmv CO limit was chosen by EPA as indicative of steady state efficient combustion based upon EPA evaluation of CO emissions from hazardous waste incinerators (56FR7151, middle column). The choice of 20 ppmv HC as protective of human health which was also based upon risk assessments conducted for 30 incinerators (56FR7155, middle column). The problem is that any similarity between incinerators and cement kilns is purely superficial.
The EPA Hazardous Waste Incinerator Model As Compared to a Cement Kiln
Throughout EPA guidance documents, background documents and research
papers, a variety of incinerator models are used. Figure 1 provides a
very generalized schematic flow diagram of a rotary kiln incinerator,
while Figure 2 provides a very generalized schematic flow diagram of a
cement kiln. Features of incinerators which are critically different
from cement kilns and can impact emissions are noted in Table I.
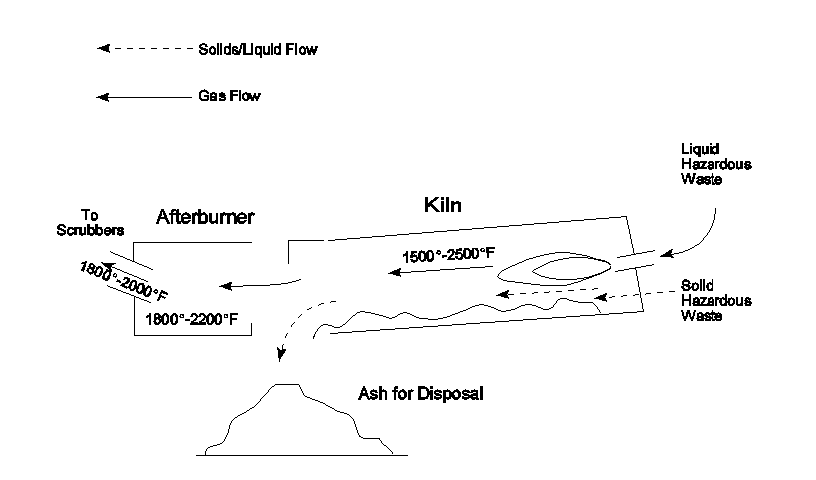
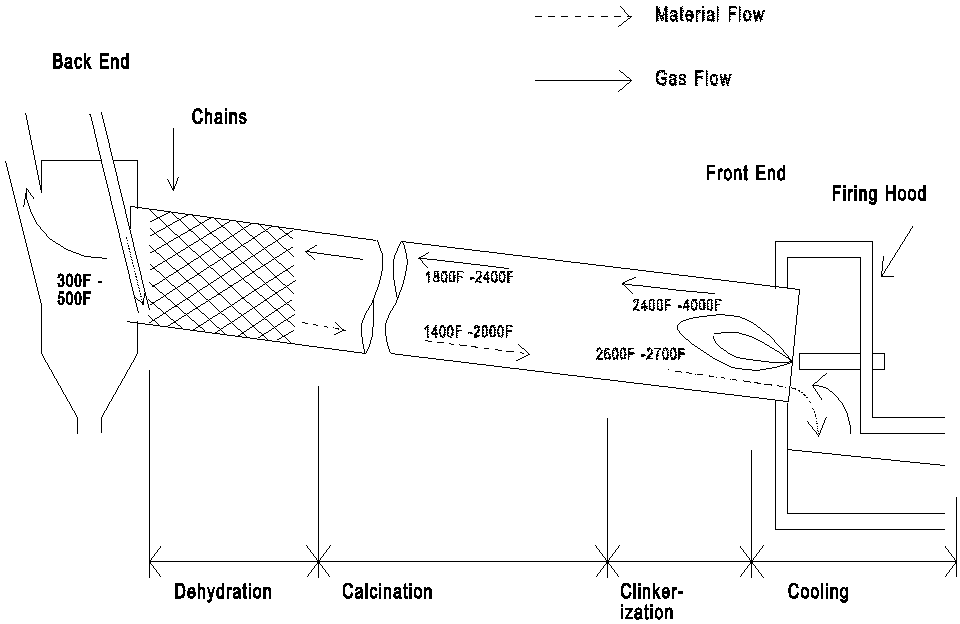
Clearly the similarities between how a hazardous waste incinerator
works and how a cement kiln works are superficial and cannot be the
basis for evaluation or regulatory controls.
Cement kilns are referenced in Co-Firing Hazardous Waste Fuels in Industrial Processes -- The Business Decision by Donald J. Henz, P.E. of PEI Associates, as providing "... the retention time, turbulence, and temperatures (the three T's) necessary to oxidize completely ..." hazardous waste fuel.
|
Incinerator |
Cement Kiln |
|
| There are no chemical reactions in an incinerator other than those induced directly by incineration (i.e. oxidation) Therefore, gases exiting the incinerator directly reflect what is happening in the combustion zone of the kiln. | In a cement kiln, the dehydration and calcination steps produce large quantities of gases that largely mask gaseous emissions from the combustion zone of the cement kiln. Nearly half the mass of raw materials that enter a wet process cement kiln leave the kiln as gases - primarily water vapor and carbon dioxide - yet they are not combustion by-products. | |
| In an incinerator the gases are moving the same direction as solids and liquids. This tends to drive any phase equilibrium to the gaseous side of the equation thus increasing emissions such as metals. Vapor pressure becomes the critical factor. | In a cement kiln, the counter current flow design tends to entrain metals in the cement clinker via the development of recirculating loads. Only if metals reach the cooler chain section prior to condensation are they likely to be enriched in the kiln emissions and even then primarily as a particulate because of the relatively low exit temperatures. | |
| The thermal capacity and therefore thermal stability of an incinerator is relatively low. Process upsets can occur within a matter of minutes or even seconds that can allow uncombusted organics to escape from the process. The use of an afterburner is largely prompted by this potential. | In a cement kiln at any given moment there is typically greater than 1,000 times as much process solids undergoing chemical reactions at 2,000 degrees F as there is hazardous waste fuel being combusted. This provides enormous thermal stability in the kiln. It would take several hours to feel the effects of a kiln upset, which generally reflects a small decrease in production capacity rather than any changes which might affect combustion. |
The benefit of "The high temperatures, long residence times, and high degree of turbulence...." of cement kilns is also referenced in Handbook of Hazardous Waste Incineration by Calvin Brunner, P.E. Additionally, since EPA has made the comparison between incinerators and cement kilns, Bob Mournighan of the USEPA has stated in his "Effect of Burning Hazardous Waste in Cement Kiln on Conventional Pollutant Emissions" that "...kiln temperatures and gas residence times are well in excess of that seen in most hazardous waste incinerators...." Cement kilns provide ample time, temperature and turbulence necessary to be ideal hazardous waste combustors and EPA has cited these types of controls (retention times and temperatures) for PCB incineration (40CFR761.70).
THE 100PPMV CO LIMIT
As mentioned previously, EPA has stated in the BIF preamble that "The 100ppmv limit...is indicative of steady state (i.e. normal), efficient combustion conditions." (56FR7151, middle column) This is, unfortunately, based upon hazardous waste incinerator data rather than relevant cement kiln data. In addition to the fact that the 100ppmv limit was established based upon incinerator data, the Agency later acknowledges that "...EPA has not established a direct correlation between CO emissions, PIC emissions and health risks." (56FR7153, middle column)
The agency goes on to justify the 100ppmv CO position by stating
that "...the 100ppmv CO limit is reasonable for a number of reasons.
Not only is it within the range of CO levels that are indicative of
good combustion conditions, but the Agency believes that it is not too
low because: (1) It is higher than the technology based 50 ppmv CO
level EPA requires for boilers burning waste PCBs (see 40CFR761); (2)
it is higher than the CO limits included in many hazardous waste
incinerators permits; (3) the Agency explicitly encourages the use of
pre-alarms to minimize the frequency of automatic waste feed cut-offs;
and (4) the limit is implemented on an hourly rolling average basis
which allows and minimizes the effects of short-term CO spikes." So,
not only did EPA base the 100ppmv CO limit on incomparable incinerator
data, technology based boiler data is also referenced. While those CO
limits may be relevant to incinerators and boilers, CO as a combustion
indicator in cement kilns is of questionable relevance.
Naturally Occurring Variability in CO
Elevated CO levels occur in cement kilns for a number of reasons. Calcination, which is critical to the cement production process, results in CO byproducts, as does the decomposition of carbon dioxide due to the extremely high temperatures in a cement kiln. Decomposition of carbon dioxide may also occur due to the presence of high moisture and metallic catalysts. Finally, partial oxidation of hydrocarbons present in raw materials may also produce CO. All of these examples are independent of good combustion conditions.
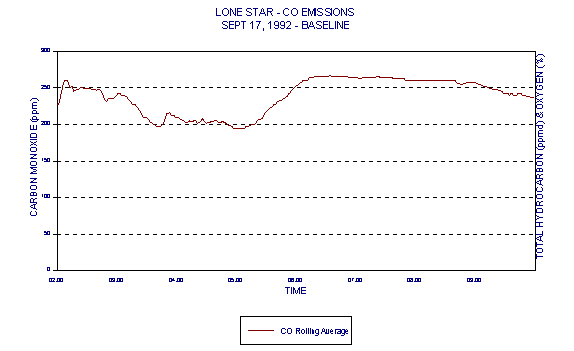
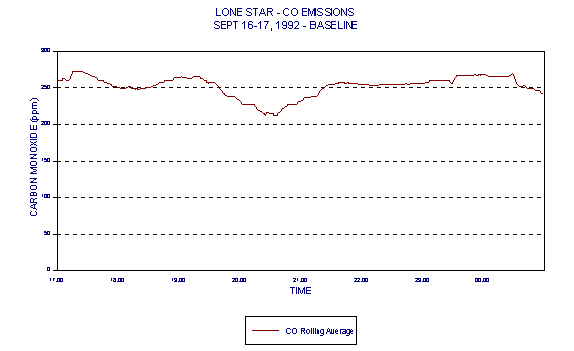
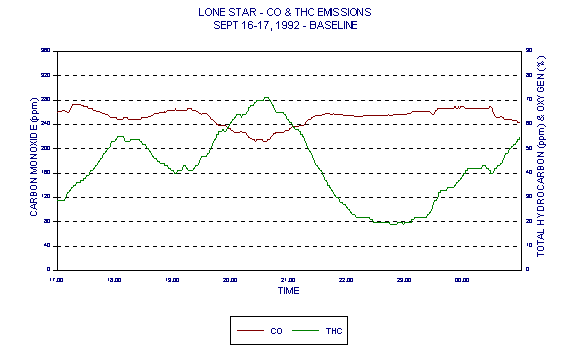
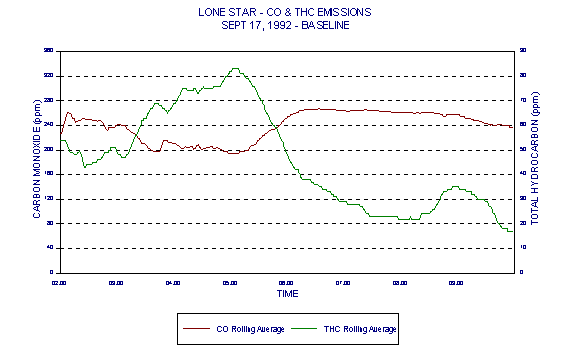
Figures 3 and 4 show levels for Lone Star Industries, Inc. Greencastle, IN facility baseline (coal only) tests conducted September 16-17, 1992 representing two eight hour test periods. The baseline tests were extended to eight hours in anticipation of demonstrating naturally occurring variability in CO levels under the normal operating conditions of a stable kiln. These graphs clearly demonstrate a wide variability in CO levels.
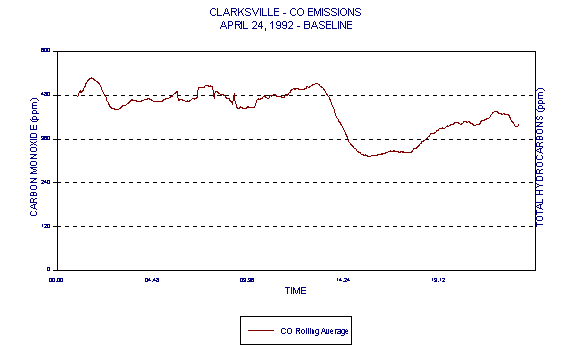
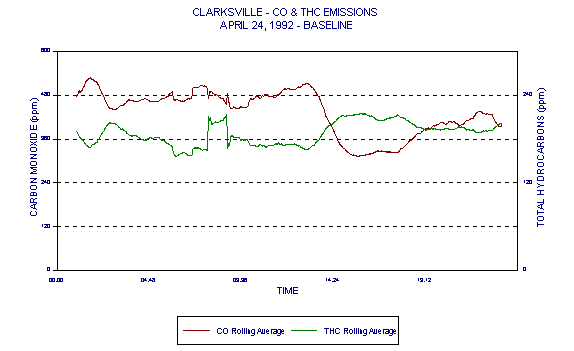
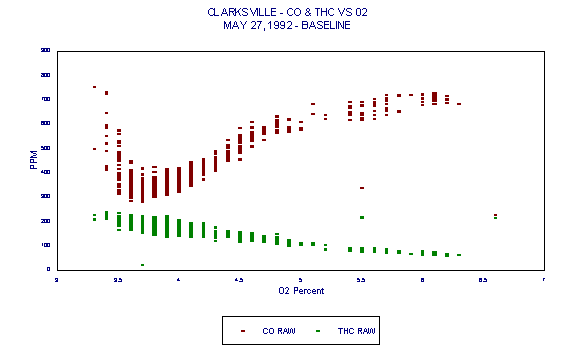
Figure 5 represents CO data measured during a baseline (coal/coke only) day of Holnam's Clarksville, MO facility Compliance Test. It is quite evident in this case that there is a wide variability in CO. This data and its variability are typical of cement kilns under both baseline and HWF use conditions. Naturally occurring variability in CO levels, due to the cement manufacturing process, prevents CO from being a reliable indicator of combustion in cement kilns.
This data, demonstrating the naturally occurring variability in CO levels, brings into question the relevance of the 100ppmv CO limit in cement kilns. Because of the variability in CO, any resulting average baseline (based upon current BIF methodologies) would also be highly variable. Monitoring baseline conditions that are so highly variable does not discriminate between good and bad combustion as intended by BIF.
HC as an Alternative to CO
In the BIF Preamble, EPA acknowledged that "...some cement kilns...routinely emit CO above the Tier I 100ppmv limit." EPA also recognized that "...massive amounts of feedstocks...can generate large quantities of CO emissions which are unrelated to the combustion efficiency of burning the waste and fuel." EPA consequently developed Tier II limits whereby "...CO levels can exceed 100ppmv provided that the owner or operator demonstrate that the HC concentration in the stack gas does not exceed a good operating practice based limit of 20 ppmv...." (56FR7153, right-hand column, Tier II PIC Controls: Limits on CO and HC). The intent of the Tier II option then is to provide a way that HC can be used as an alternative to CO.
There are some obvious assumptions here which have proven to be incorrect. First, detection of HC above a certain level is not necessarily an indication of combustion conditions, good or bad. Second, HC and CO do not exhibit a direct relationship. So, even if CO does have some bearing on combustion conditions in the cement kiln, the following discussion demonstrates that HC is not an indicator of combustion conditions above that arbitrary 100ppmv CO level. Consequently, even though the Tier II option is designed to provide an alternative to cement kilns that regularly exceed 100ppmv CO, it does not. First, consider naturally occurring HC in raw materials.
HC From Raw Materials
Many naturally occurring raw materials used in the cement manufacturing process contain significant quantities of hydrocarbons. A wet kiln slurry is typically composed of shale, limestone and water and is fed into the kiln at near ambient temperatures. As the material travels downward through the kiln, the material temperatures rise. A portion of the hydrocarbons contained in the raw materials are vaporized and pyrolyzed as temperatures increase. Organic compounds (hydrocarbons) are swept out of the kiln with the countercurrent gases.
Table 2 shows total organic carbon (TOC) tests performed on the raw
feed during Holnam's Clarksville, MO facility Compliance Test. This
data varies significantly from sample to sample. No correlation has
been established between total organic carbon test results and
hydrocarbon emissions. Differences in the types of hydrocarbons present
as well as factors such as kiln temperature profile and oxygen
conditions are the true independent variables in this case, but they
are very difficult to correlate with HC emissions. It is also of
interest to note that temperature profiles within a kiln, along with HC
and CO emission levels, have been shown to change even with
non-hazardous alternate fuel sources, such as diesel fuel.
|
Compliance Test Date |
ppm |
|
April 21, 1992 |
5,060 |
|
April 22, 1992 |
2,900 |
|
April 23, 1992 |
13,100 |
|
April 24, 1992 |
15,600 |
Organic materials entering the kiln via the raw feed slurry were also measured at Continental Cement Company's Hannibal, MO facility. The high level of organics in the raw material is evident in the data presented in Table III. A wide variation was also measured. This data was collected over the course of a week.
TOC was measured for all four Compliance Test days for the Lone Star
Industries, Inc. Greencastle, IN facility. This data is presented in
Table IV. Note the level of organics as well as the wide variation, as
was evident at both Clarksville and Continental.
|
|
Organic Input |
|
Test Day 1 |
182 |
|
Test Day 2 |
815 |
|
Test Day 3 |
910 |
|
Test Day 4 |
1,542 |
|
Test Day 5 |
567 |
Table IV. Lone Star Compliance Test Slurry TOC.
|
|
ppm |
|
May 18, 1992 |
1,750 |
|
May 19, 1992 |
3,400 |
|
May 20, 1992 |
6,200 |
|
May 21, 1992 |
2,240 |
Clearly, the level of naturally occurring hydrocarbons found in the raw materials at each of these cement plants presents a significant source of hydrocarbons in the kiln. As mentioned earlier, these hydrocarbons are driven off as the slurry is heated, before it ever reaches the combustion zone. This is a constant source of measurable stack HC, which is highly variable and completely unrelated to fuel combustion.
Variability of HC
Figures 3 through 5 demonstrated variability of CO. Figures 6 through 8 are those same graphs with HC plotted also. The variability of HC is evident. But note in particular that not only do HC and CO exhibit significant variability over time, they also exhibit an inverse relationship. The inverse relationship alone precludes any usefulness of HC as an alternative combustion indicator to CO as intended in the BIF regulations.
In addition, this inverse relationship is not always consistent. It is thought that at times, HC entering with raw materials is partially oxidized in the presence of additional oxygen. This partial oxidation leads to an increase in CO and a decrease in HC. This results in the occasional inverse relationship between HC and CO.
Additional Unsuitability of HC as a Combustion Indicator
Figure 9 illustrates a test performed at Holnam's Clarksville, MO facility when the O2 content was varied from a very low concentration where it was generating high CO, to a very high O2
concentration. It was observed that a minimum O2 concentration was exhibited beyond which a further decrease in the O2 concentration increased CO dramatically with essentially no change in HC. These low oxygen conditions clearly show a significant increase in CO, yet little or no effect on HC.
Low oxygen conditions fairly consistently result in elevated CO levels. This indicated some type of relationship between poor combustion conditions, such as low oxygen, and some level of elevated CO. This trend warrants further examination. Anticipate that this relationship will likely be kiln specific.
The data for Figure 10 was generated at Continental Cement Company's Hannibal, MO facility during a test in which the kiln was purposely operated under conditions of poor combustion to observe the effect on HC emissions. This phenomenon is also illustrated in Figures 11 and 12. Even under poor combustion conditions, the effect on HC concentrations was of doubtful statistical significance.
The data presented in this document, demonstrates the unsuitability
of CO or HC as reliable combustion indicators utilizing existing
standards. Under good combustion conditions, the natural variability
factor clearly shows why these parameters are not reliable combustion
indicators. The all too frequently observed inverse relationship
complicates matters beyond practicality.
Given that CO and HC, as currently utilized, are not reliable combustion indicators under baseline conditions, how can the measurement of these parameters be relevant to combustion while burning hazardous waste? The answer is that CO and HC are not reliable combustion indicators with or without burning hazardous waste fuels. How then can combustion conditions be monitored in order to insure the prevention of adverse effects on public health and the environment?
ALTERNATIVE OPERATING LIMITS
Clearly, the existing HC and CO limits are not good combustion indicators in cement kilns. There are too many interferences for these parameters to even be considered conservative indicators. In order to insure proper combustion in the kiln while hazardous waste fuel is being burned, limits should be established for a minimum oxygen level at the kiln exit and a minimum combustion zone temperature. This will insure that sufficient oxygen is available for combustion, and sufficient temperature is maintained to destroy the wastes. Kiln exit oxygen should be limited to a minimum hourly rolling average.
In addition, limit the combustion zone temperature where measurement is as close to the combustion zone as possible. Automatic waste feed cut-offs would be established based upon those parameters so that hazardous waste fuel will be automatically shut off if those limits are not met.
One specific area that merits additional investigation concerns kiln
exit oxygen and true combustion related carbon monoxide. Air leakage
into the kiln, between the kiln exit and the stack, results in stack
oxygen levels that vary considerably and do not reflect kiln combustion
conditions except on a relative and short term basis. Figures 9, 11 and
12 also clearly show that combustion related CO is produced at low
oxygen levels. Nevertheless, CO under current rules for establishing a
baseline is a poor indicator because of previously discussed reasons.
Kiln exit oxygen, which does represent combustion conditions, has been
suggested as an alternate monitoring parameter. It is proposed that on
different kilns, the relationship between CO and kiln exit oxygen be
studied under various operating conditions in order to potentially
establish a minimum kiln exit oxygen level which insures complete
combustion under a variety of cement kiln operating conditions.
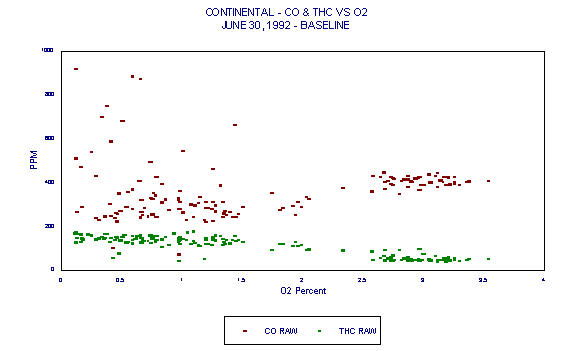
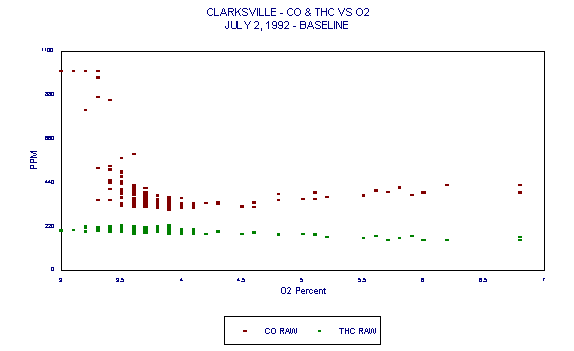
Figure 12. Clarksville CO & THC vs O2 July 2, 1992 - Baseline.
CONCLUSION
In numerous tests it becomes increasingly obvious that THC and CO are not suitable combustion indicators. THC and CO have actually been demonstrated to sometimes increase when hazardous waste fuels are not being burned. In addition, HC and CO have been shown to, at times, have an inverse relationship. Clearly, pursuit of the use of THC and CO as combustion indicators in cement kilns is unlikely to be successful.
The following issues need to be reconciled:
- Cement kilns and incinerators are significantly different devices. Why then are current HC and CO limits based upon incinerator data?
- Elevated CO levels have been demonstrated to occur in most cement kilns independent of combustion conditions. What then is the relevance of setting a maximum CO level?
- HC emissions occur because of natural hydrocarbon sources (i.e. kerogens) in raw materials which are unrelated to combustion conditions. Why then are hydrocarbons thought to be good combustion indicators?
- HC and CO levels vary significantly, a phenomenon that is unrelated to combustion conditions. How then can baselines be established, and what is the relevance?
- HC cannot be used as an alternative to CO because tests under starved oxygen conditions have demonstrated negligible effects on HC. Monitoring HC fails to detect poor combustion conditions.
The establishment of controls to insure complete combustion in
cement kilns must be developed based on how cement kilns operate, not
incinerators. Using cement kiln based operating parameters will help
ensure that controls are developed which are truly protective of human
health and the environment.
References
1. A. Beers, "Hazardous Waste Incineration: The Cement Kiln Option," New York State Legislative Commission on Toxic Substances and Hazardous Wastes, 1987.
2. P.E. Brunner , R. Calvin, Handbook of Hazardous Waste Incineration, Blue Ridge Summit, Tab Books, Inc. PA, 1989.
3. D.G. Gossman, J. Chapman et al., "Trial Burn and Compliance Test Report For Continental Cement Company", Volumes 1-6, Gossman Consulting, Inc., Hampshire IL, 1992.
4. Engineering-Science, "Background Information Document for the Development of Regulations to Control the Burning of Hazardous Wastes in Boilers and Industrial Furnaces, Volume II," draft manuscript, US EPA, Washington DC, 1987.
5. Environmental Protection Agency, "Burning of Hazardous Waste in Boilers and Industrial Furnaces - Supplement to Proposed Rule", Federal Register, Volume 54, No. 206, US Government Printing Office, Washington DC, 1988, pp 43717-43763.
6. Environmental Protection Agency, "Burning of Hazardous Waste in Boilers and Industrial Furnaces; Proposed Rule," Federal Register, 1987.
7. Environmental Protection Agency, "Burning of Hazardous Waste in Boilers and Industrial Furnaces - Final Rule", Federal Register, Volume 56, No. 35, US Government Printing Office, Washington DC, 1991, pp 7134-7240.
8. Environmental Protection Agency - Region 4, "Boiler and Industrial Furnace Regulations and Implementing Metals/HCl and PIC Controls Under Omnibus Authority", Meeting on Proposed Changes in Incinerator, 1988.
9. D. G. Gossman, J. Woodford et al., "Trial Burn and Compliance Test Report For Holnam Inc. - Clarksville Cement Plant", Volumes 1-6, Gossman Consulting, Inc., Hampshire IL, 1992.
10. D.G. Gossman, J. Zuzulo et al., "Trial Burn and Compliance Test Report For Lone Star Industries, Inc.", Volumes 1-6, 1992.
11. D.G. Gossman, Review of New EPA Proposed Regulations on Organic Emissions From Cement Kilns Burning Hazardous Waste Fuels, Gossman Consulting, Inc., Hampshire IL, 1989.
12. P.E. Henz, D.J., "Co-Firing Hazardous Waste Fuels in Industrial Processes -- The Business Decision", in Proceedings of the 79th Annual Meeting, the Air Pollution Control Association, Minneapolis MN, 1986.
13. R.E. Mournighan, J. A. Peters et al., "Effects of Burning Hazardous Waste in Cement Kilns on Conventional Pollutant Emissions," in Proceedings of the 78th Annual Meeting, Air Pollution Control Association, Detroit MI, 1985.
14. D.G. Gossman, The Reuse of Petroleum and Petrochemical Waste
in Cement Kilns, Gossman Consulting, Inc., Hampshire IL, 1991.